Global Blood Collection Tube for Liquid Biopsy Market Surges with Europe’s Leadership; QIAGEN N.V., F. Hoffmann-La Roche
Advanced Diagnostics, Precision Medicine Adoption, and Regulatory Alignment Accelerate Market Growth Through 2035
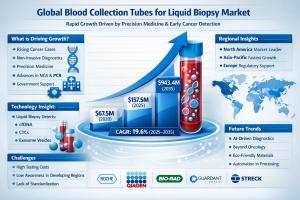
ROCKVILLE, MD, UNITED STATES, April 10, 2026 /EINPresswire.com/ -- According to the latest analysis by Fact.MR, the global blood collection tube for liquid biopsy market is valued at USD 157.5 million in 2025 and is projected to reach USD 188.4 million in 2026, before surging to USD 943.4 million by 2035, expanding at a robust CAGR of 19.6%. The market is expected to create an incremental opportunity of over USD 785 million during the forecast period.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=4495
The transformation of this market is being driven by the rapid shift toward minimally invasive diagnostics, rising cancer prevalence, and increasing integration of precision medicine frameworks. Liquid biopsy technologies enabled by advanced blood collection tubes are redefining early detection, treatment monitoring, and personalized care pathways.
Quick Stats
Market Size (2025): USD 157.5 Million
Market Size (2026): USD 188.4 Million
Forecast Value (2035): USD 943.4 Million
CAGR (2025–2035): 19.6%
Incremental Opportunity: ~USD 785 Million
Leading Segment: CTC (Circulating Tumor Cell) Tubes
Leading Region: Europe (Germany as key hub)
Key Players: QIAGEN N.V., F. Hoffmann-La Roche Ltd., Biocept Inc., Streck Inc., Greiner Bio-One International GmbH
Executive Insight for Decision Makers
The market is undergoing a strategic shift from conventional biopsy methods to real-time, non-invasive diagnostics. Germany, as a European innovation hub, is at the forefront due to strong regulatory alignment under IVDR and robust oncology research infrastructure.
What stakeholders must do:
Invest in NGS-compatible and AI-integrated collection systems
Strengthen regulatory compliance capabilities across Europe
Build strategic alliances with diagnostic labs and biotech firms
Risk of inaction:
Companies failing to adapt risk losing relevance in precision diagnostics ecosystems, where speed, accuracy, and compliance define competitiveness.
Market Dynamics
Key Growth Drivers
Rising global cancer incidence and demand for early detection
Expansion of precision medicine and personalized therapies
Technological advancements in cfDNA, CTC, and EV stabilization
Government funding and regulatory support for non-invasive diagnostics
Key Restraints
High cost of liquid biopsy testing
Lack of standardized protocols in emerging markets
Limited awareness in developing regions
Emerging Trends
AI-driven biomarker analysis and automation
Expansion beyond oncology into neurology and infectious diseases
Development of eco-friendly and biodegradable tubes
Integration with NGS and PCR platforms
Segment Analysis
Leading Segment: CTC tubes dominate with over 40% market share, driven by oncology applications
Fastest-Growing Segment: cfDNA preservation tubes due to increasing use in multi-cancer early detection
Breakdown:
Product: CTC tubes, cfDNA tubes, RNA stabilization tubes
Application: Cancer diagnosis & monitoring (largest), NIPT, infectious diseases
End-use: Hospitals & diagnostic labs (dominant), research institutes
Strategic Importance:
Oncology-driven segments remain revenue anchors, while multi-disease applications unlock future scalability.
Supply Chain Analysis (Critical Insight)
Raw Material Suppliers:
Specialty chemical providers (preservatives, anticoagulants)
Polymer and medical-grade plastic manufacturers
Manufacturers / Producers:
Tube manufacturers such as Greiner Bio-One International GmbH and Streck Inc.
Distributors:
Medical device distributors
Laboratory supply chains across Europe
End-Users:
Hospitals (cancer diagnostics)
Diagnostic laboratories (high-volume testing)
Research institutions (clinical trials, biomarker research)
Who supplies whom:
Raw material suppliers → Tube manufacturers → Diagnostic distributors → Hospitals & labs → Patients
Germany plays a central role with localized manufacturing and strong distribution networks across the EU.
Pricing Trends
Commodity vs Premium:
Standard tubes: Price-sensitive
Advanced preservation tubes: Premium pricing
Key Influencing Factors:
Raw material quality and preservatives
Regulatory certifications (CE, IVDR compliance)
Compatibility with NGS/PCR platforms
Margin Insights:
Premium tubes offer higher margins (20–35%)
Commodity products face margin pressure due to competition
Regional Analysis
Top 5 Countries by CAGR (2025–2035):
Germany – ~18.5%
United States – 20.7%
China – 19.2%
Japan – 16.6%
United Kingdom – ~17.8%
Why Germany Leads:
Strong biotech ecosystem
Advanced healthcare infrastructure
Early adoption of IVDR regulations
Developed vs Emerging Markets:
Developed: High adoption, strong compliance
Emerging: High growth potential, cost-sensitive
Competitive Landscape
Market Structure: Moderately fragmented
Top 5 Players: Control ~45–55% market share
Key Companies:
QIAGEN N.V.
F. Hoffmann-La Roche Ltd.
Biocept Inc.
Norgen Biotek Corp.
Greiner Bio-One International GmbH
Competitive Strategies:
Product innovation (stabilization technologies)
Strategic partnerships with diagnostics firms
Geographic expansion into Asia-Pacific
Regulatory approvals for market access
Strategic Takeaways
For Manufacturers
Focus on advanced stabilization and automation
Expand Europe-based production hubs
For Investors
Target companies with AI and NGS integration capabilities
Invest in multi-disease diagnostic platforms
For Marketers / Distributors
Build strong hospital and lab partnerships
Educate markets on liquid biopsy benefits
Future Outlook
The market is poised for transformational growth, with liquid biopsy expanding beyond oncology into neurology, infectious diseases, and prenatal diagnostics.
Key future drivers:
AI-powered diagnostics
Automation in sample processing
Sustainable product innovation
By 2035, blood collection tubes will become central to next-generation diagnostic ecosystems, enabling faster, more accurate, and scalable healthcare solutions.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/4495
To View Our Related Report:
Blood Cancer Diagnostics Market:https://www.factmr.com/report/47/blood-cancer-diagnostics-market
Blood Clot Retrieval Devices Market: https://www.factmr.com/report/blood-clot-retrieval-devices-market
Blood Sugar Tester Market: https://www.factmr.com/report/blood-sugar-tester-market
Blood Gas Analyzers Market: https://www.factmr.com/report/blood-gas-analyzers-market
S. N. Jha
Fact.MR
+1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.